We study how symmetries are broken or maintained during embryonic development and growth. Why are our hearts on the left and how do they come to be there? How do our two arms develop to be the same size? How does our spine stay straight during growth, and how does this go wrong in diseases like scoliosis? These are just some of the questions we are addressing in our work.
These interests have led us into all kinds of fascinating biology including fluid flows in the embryo and brain, transduction of mechanical signals, the enigmatic Polycystin proteins, ciliated neurons, cell polarity, and more. Read on for further details.
A cilium in the embryonic node of mouse
Cilia
It turns out that organelles called cilia are critical for the making and breaking of many symmetries. For instance, they are needed for the correct left-right asymmetric position of internal organs like the heart and liver, while we recently discovered that cilia are also be required to prevent the spine from curving and kinking to the side.
Cilia come in two main flavors: 1) sensory (or primary) cilia, which are small and don't move but are very important cellular "antenna" that receive signals from the outside environment and transduce them into the cell. Sensory cilia were originally linked to hedgehog signal transduction, but it is now known that they are involved in a range of different chemical signaling pathways and also in the transduction of other kinds of signal, like light, sound, and forces. 2) The second main branch are the motile cilia. Usually longer and often in great numbers on the surface of cells, these cilia beat back and forth to generate microscopic fluid flows through tubes and across epithelial sheets.
Disruption of cilia function causes a group of diseases called ciliopathies. The primary ciliopathies include Bardet-Biedl Syndrome, Alstrom Syndrome, Joubert Syndrome, and many others. They are typically characterized by multi-organ defects. Motile cilia dysfunction, by constrast, causes Primary Ciliary Dykinesia, which is characterized by inability to clear the airways, male and female infertility, and sometimes abnormal left-right patterning.
How cilia break left-right symmetry of the embryo
The symmetry of many vertebrate embryos is broken by a mechanism that involves both motile and primary cilia, described in detail in our recent review. During early somite stages, cilia exist within a midline left-right organizer (LRO) - the node in mouse and Kupffer's vesicle in zebrafish. Many of these cilia are motile and they generate an extracellular fluid flow that is left-right asymmetric. Somehow this flow is sensed by primary cilia, also within the LRO, and, as a result, asymmetries in gene expression emerge first around the LRO and then more laterally in the lateral plate mesoderm. Ultimately, gene expression asymmetries - with Nodal and its downstream targets being expressed in the left but not right lateral plate mesoderm - direct the asymmetric morphogenesis of the organs. We are interested in several aspects of this process:
1. What are the molecular mechanisms that lead to formation of motile cilia and asymmetric fluid flow in LROs?
2. How is fluid flow sensed? Our current working hypothesis is that Polycystin proteins, localized to primary cilia, are involved in a mechanosensory response to the force of fluid flow. We aim to understand this mechanotransduction mechanism and the downstream signaling events that result in gene expression asymmetries.
3. How is the embryo asymmetrically patterned? Left-right patterning involves the spatially and temporally controlled interactions of many secreted factors - activators and inhibitors of Nodal signaling. We are interested in understanding how these factors move in the embryo and interact with one another to establish robust left side-only activation of the Nodal pathway.
Credit: joanthon rosen
How cilia maintain the symmetry of the spine
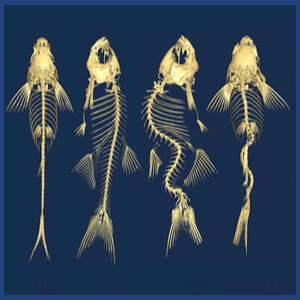
We know surprisingly little about how the spine remains straight during its growth. Idiopathic Scoliosis, a common disease impacting 3-4% of the worlds adolescents, involves 3D curvatures and rotations of the spine but we do not understand the underlying causes.
We found that mutants that effect motile cilia function in zebrafish cause late-onset 3D spinal curves to develop. Motile cilia in the brain ventricles and spinal canal are important for generating cerebrospinal fluid flow, and so we hypothesize that cilia-driven flows in the brain and spine are somehow critical for maintaining spinal symmetry. In this project, our main interests lie in:
1. Understanding the cellular and molecular implications of defective cerebrospinal fluid flow and deciphering how these give rise to spinal curves.
2. Utilizing modern genome editing technologies to develop zebrafish models of Idiopathic Scoliosis for investigations of the underlying causes of this disease.